GOP senators call for restrictions on generic abortion drugs
Fifty-one U.S. Senators called on the U.S. Food and Drug Administration and the Department of Health and Human Services on Thursday for more restrictions on access to abortion drugs, including a recently-approved generic drug.
In a letter to agency heads, led by Sen. Lindsey Graham, R-S.C., the Republican senators said mail-order access without first visiting a doctor puts women and children at risk. The letter comes after the FDA quietly approved a generic version of mifepristone last week.
The Senators said the FDA’s move also undercuts the anti-abortion movement.
“Unrestricted access to abortion pills is systematically undermining states’ rights and violating pro-life state laws,” senators wrote. “Every month, thousands of abortion drugs are shipped into states that have otherwise limited access to abortion after Dobbs, degrading pro-life laws at the state level across the country.”
The letter was sent to Health and Human Services Secretary Robert F. Kennedy Jr. and FDA Commissioner Marty Makary on Thursday.
Marjorie Dannenfelser, president of Susan B. Anthony Pro-Life America, said the FDA must protect children.
“At a minimum the FDA must immediately reverse the Biden-era policy of removing doctors by allowing mail-order abortion drugs without in-person medical oversight,” she said in a statement. “Women are endangered by taking the drugs without a sonogram to check for ectopic pregnancy and confirm the gestational age of the child.”
The senators also called for a change in culture in the letter.
“The ‘abortion-on-demand’ culture enabled by the Biden-Harris administration’s removal of critical safeguards on the only FDA-approved abortion regimen is currently the biggest threat to unborn life in America today,” they wrote. “Today, your agencies have all the information they need to bring an end to previous Democrat-administrations’ abortion drug regulations while a comprehensive review is conducted.”
Senators called for five immediate actions:
Suspend the approval of any new generic versions of mifepristone pending the outcome of the REMS safety review;Commit to ensuring that all generic versions of mifepristone are included in the ongoing reevaluation of prior REMS approvals;Reinstate the in-person dispensing requirement for mifepristone and all its generic versions;Suspend the distribution of mifepristone and all generic versions as an “imminent hazard” under Section 505(e) of the Federal Food, Drug, and Cosmetic Act (FDCA); andWithdraw FDA guidance permitting pharmacy distribution of mifepristone and all its generic versions.
Last week, the FDA approved a generic form of mifepristone, one of the two medications used in most U.S. abortions. Mifepristone is typically used alongside misoprostol to induce abortions. Medication abortions account for 63% of abortions in the U.S., according to a report from the Guttmacher Institute.
HHS Communications Director Andrew Nixon previously told The Center Square that “the FDA has very limited discretion in deciding whether to approve a generic drug.”
“By law, the Secretary of Health and Human Services must approve an application if it demonstrates that the generic drug is identical to the brand-name drug,” Nixon said.
Latest News Stories
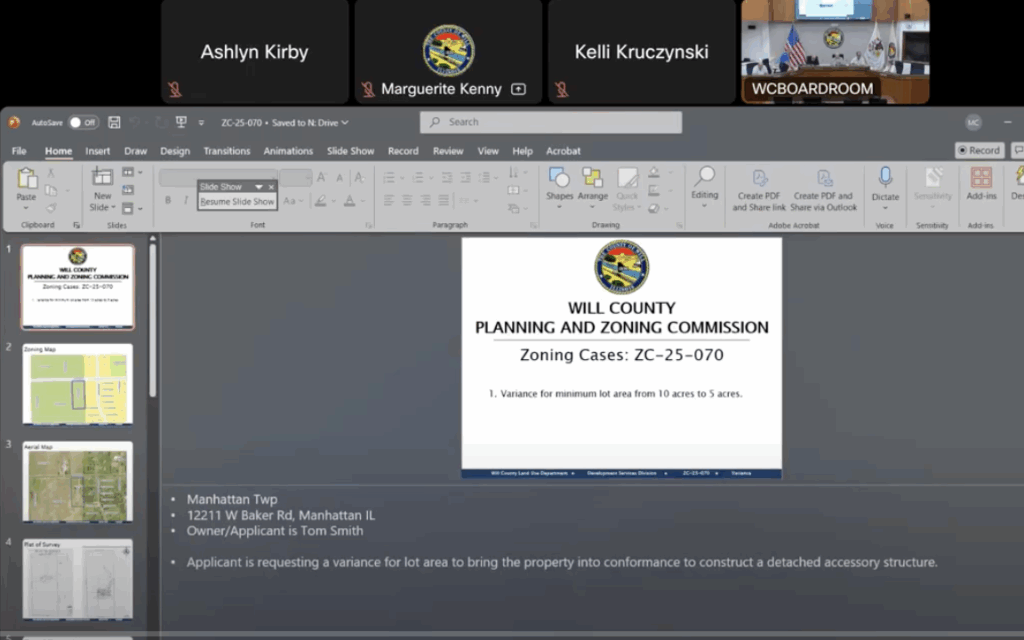
Will County Planners OK Oversized Garage Near Naperville, Overriding Staff Recommendation

Green Garden Landscaping Business Gains Permit Amid Strong Neighbor Support

Meeting Briefs: Will County Planning and Zoning Commission for July 15, 2025

Frankfort Approves ‘Whisk & Flame’ Culinary Studio, Slashes Parking Requirement for Downtown Property

Frankfort Village Board Adopts $59.4 Million Appropriation for Fiscal Year 2026

Frankfort Establishes New Zoning Rules to Attract Data Centers

Currie Motors Expansion Gets Approval with Site Modifications

Frankfort Approves $134,531 Maintenance Contract for Wastewater Plant Filters

Meeting Briefs: Frankfort Village Board for July 14, 2025

County Approves School Resource Officer, Multi-Year Planning Requirements

County Addresses Senior Tax Exemption Processing Error

Executive Committee Meeting July 10 Meeting Briefs